How to Read a Dsc Curve Plastic
Abstruse
This work is about the identification of polymers by means of differential scanning calorimetry (DSC), thermogravimetry (TG) and simultaneous thermal analysis (STA) involving computer-assisted database search. One full general limitation depicted is the possibility of multiple interpretations of a single measurement bespeak which sometimes makes definite identification difficult. It is shown that a consecutive but also simultaneous incorporation of two types of measurements can significantly reduce multiple interpretations and thus increment the odds of right identification. The latter is furthermore enhanced past using the recently introduced KIMW database which contains DSC curves of 600 different commercially available polymers (about 130 polymer types) including data about trade names, colors and filler contents.
Introduction
The identification of materials such every bit those from the field of polymers is of slap-up interest, particularly with regard to the quality control of raw materials and the failure analysis of entire building elements [i, 2]. Measurement techniques like adulterate full reflectance Fourier transform infrared spectroscopy (ATR) tin can for case be applied to characterize the composition of polymers [iii]. The most common methods of classical thermal analysis such as differential scanning calorimetry (DSC) and thermogravimetric analysis (TG) are likewise widely used in guild to investigate polymers [4, 5]. Caloric effects observed in the DSC betoken, e.one thousand., the glass transition, crystallization and melting, equally well as the pyrolytic decomposition and the combustion of the polymer samples studied by means of TG, allow for a detailed characterization. There are furthermore avant-garde DSC tests like the determination of the oxidation induction time (OIT) which provides data nigh the thermal stability of polymers [1, 5]. It should be noted that in full general two modes of DSC can be distinguished [ane, 4, 6]: estrus flux versus power bounty; in this work, exclusively heat flux DSC was utilized which should non exist put on a level with the simpler DTA (differential thermal analysis) method [6]. Simultaneous thermal assay (STA) refers in general to the application of two or more techniques to the aforementioned sample at the same time [6]; in this piece of work, STA signifies the simultaneous measurement of DSC and TG. The STA technique plain has important advantages over only combining measurements performed in different instruments on different samples of the same type: Besides the higher efficiency, the TG and DSC signals from STA measurements can exist compared directly without any discussion about possibly different sample compositions, sample preparations or measurement weather condition. Stand-alone TG instruments may also offer the possibility of a semiquantitative detection of caloric effects via c-DTA ® which is a calculated differential thermal analysis (DTA) curve [6,7,8]. The latter is evaluated from the difference betwixt the temperature–time bend during the sample measurement and the temperature–time curve where no sample is measured. Both TG and STA instruments are oftentimes coupled to evolved gas assay techniques such as mass spectrometry (MS) or Fourier transform infrared (FT-IR) spectroscopy in order to enhance the possibilities for a material characterization [9]. Such coupled instruments are, however, not in the scope of this work.
The curve recognition and database arrangement for thermal analysis, called Identify, was launched for DSC measurements on polymers [x,11,12,xiii]. A substantial expansion of Identify was introduced recently implementing data besides from TG, dilatometry (DIL), thermomechanical analysis (TMA) and the specific heat capacity c p within the aforementioned database system [14]. The database—which can be expanded by users—includes currently more than 1100 measurements and literature data from the fields of polymers, organics, food and pharma, ceramics and inorganics, metals and alloys too as chemical elements. The latest expansion of the database is the recently introduced, optional KIMW [fifteen] library; it contains the DSC curves of 600 different commercially available polymers (most 130 polymer types) including information about trade names, colors and filler contents [16].
For a comparison of a measurement bend with thermoanalytical literature data, printed collections of results were utilized in the past, as published earlier [17, 18]. Furthermore, online databases containing thermoanalytical data [19, 20] were established already a long time earlier the launch of Place. All the same, Identify is notwithstanding unique because it is significantly different in many aspects and offers possibilities far beyond existing online databases [14]: Identify is the only bend recognition system in thermal analysis, especially when AutoEvaluation of the measurement is involved. It incorporates avant-garde event-based as well every bit datapoint-based algorithms that can be adjusted for such instances equally single- or multicomponent samples. Place allows not but for ane-on-one comparisons between measurements, only as well for nomenclature versus groups containing measurements and literature data. 1 of the biggest advantages of Place is probably the option of simply overlaying the actual measurement with database curves—fifty-fifty of a dissimilar measurement type.
This work focuses on the possibilities regarding the identification of polymers using differential scanning calorimetry, thermogravimetry and simultaneous thermal analysis in combination with a computer-assisted database search applying Place. A general limitation regarding the identification of materials via DSC and TG is the known dependence of the measurement curves on measurement conditions such as the heating rate, the sample mass or the type of crucible and lid [fourteen]. Therefore, only measurements with similar measurement conditions should ideally be considered for comparisons which can be achieved past filtering of the database [14]. Another fundamental limitation of this method of material identification is that sometimes multiple interpretations of a single measurement signal are possible [14]. This state of affairs is significantly improved past the main innovation of this work, which is the consecutive but also simultaneous incorporation of two types of measurements into the database search—as was announced in Ref. [14]. The do good of the KIMW [15] library with DSC curves of 600 different polymers [16] is furthermore shown.
Experimental
The differential scanning calorimetry measurements shown as well as other DSC database measurements were performed using the NETZSCH DSC 214 Polyma, aluminum crucibles (blazon Concavus) with pierced lids, and pure nitrogen every bit the purge gas at a period charge per unit of 40 mL min−1. In the example of all of the DSC measurements, each sample was heated at 10 K min−i to above its melting temperature, cooled down at 10 K min−ane to its individual minimum temperature and heated again at 10 K min−one to above the melting temperature. Since the second heating results are near meaningful because of a defined thermal history [1], just those curves are considered. The samples with masses typically in the range between 9 and xi mg were prepared using a SampleCutter for good thermal contact to the crucible.
The thermogravimetry measurements included in the database were carried out with the NETZSCH TG 209 F1 Libra ® using open alumina crucibles and too pure nitrogen as a purge gas at a period charge per unit of 40 mL min−1. The samples with masses of again near 10 mg were heated at 10 K min−i to 800 °C.
The measurement of a xxx% glass fiber-filled polyamide 66 sample "PA66-GF30_STA" was conducted with a NETZSCH STA 449 F3 Jupiter ® , which was equipped with a steel furnace with liquid nitrogen cooling, using platinum crucibles with pierced lids and a nitrogen purge gas menstruation of seventy mL min−1. The initial sample mass was 10.41 mg. A thermogravimetry measurement of a polybutene sample "PB_TGA_new" was performed using again the NETZSCH TG 209 F1 Libra ® under the same conditions every bit for the database measurements mentioned above; the sample mass was 10.07 mg. Withal, the temperature programme of the STA and TG measurements of the samples "PA66-GF30_STA" and "PB_TGA_new" were carried out in the same way equally for the DSC measurements (see above) where just the 2d heating results are shown and considered for the database search.
Results and discussion
Figure i shows a option of the glass transition, melting and decomposition temperatures for several polymer types [21]. It is important to emphasize that all polymer types selected exhibit exactly one drinking glass transition and one melting effect in the DSC betoken and only ane chief decomposition footstep when measured nether pyrolytic conditions via thermogravimetry. There are many other polymer types that are for case purely amorphous and do therefore not reveal a melting upshot, or types that testify several glass transitions or several decomposition steps; such polymers, which will be discussed below, are not illustrated in Fig. 1. Moreover, it must be pointed out that the characteristic temperatures shown in Fig. ane vary typically between 10 K and fifteen K when different grades of the same polymer blazon are compared. Other polymer types like thermosets not included in Fig. one showroom even much larger ranges in which these characteristic temperatures tin can exist observed.
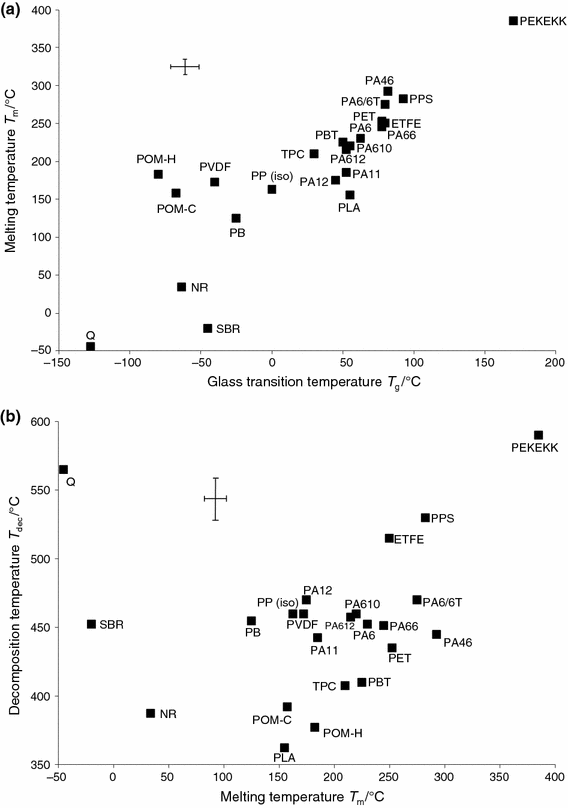
Glass transition, melting and decomposition temperatures of selected polymer types (information extracted from [21]). Simply those polymer types were selected every bit examples that showroom one glass transition and one melting issue in the DSC point and that furthermore evidence only ane main decomposition step. Additionally, these principal feature temperatures of the polymer types selected testify variations between unlike grades of i type but within a typical range which is indicated past the uncertainty bars
From Fig. 1, the known correlation between drinking glass transition and melting temperatures [22], \( T_{g}/M \approx 2/3 \;\cdot\;T_{thou}/K \), as well as betwixt melting and decomposition temperatures tin can be seen: a polymer blazon with a higher drinking glass transition temperature has by tendency a higher melting temperature and a higher decomposition temperature. Conspicuously, the polymers located at the lower and upper ends of the temperature scales in Fig. ane can be identified with greater certainty via their DSC and TG signals because there are usually less alternatives. The polymers or polymer blends that exhibit more than 1 glass transition or several decomposition steps (not illustrated in Fig. 1) can in most cases exist identified more hands on the basis of their characteristic temperatures. Identification of such polymer types was already demonstrated earlier [10,11,12,13,14, 23] and should therefore not be highlighted over again. This work focuses on more difficult cases where the glass transition temperatures are typically in the range of nearly 50–100 °C, the melting temperatures in the range of 150–250 °C or the decomposition temperatures effectually 450 °C, where various polymer types are a possibility, as tin can exist seen in Fig. one. In those cases, the consecutive or even simultaneous incorporation of DSC, TG and c-DTA ® is particularly decisive for overcoming or at least improving the situation of multiple possible interpretations [fourteen].
Incorporation of TG in addition to DSC and vice versa
Figure 2 shows the results of an STA measurement of the polymer "PA66-GF30_STA". The DSC curve exhibits a small-scale step at a mid-temperature of about 74 °C, which is due to the glass transition, every bit well every bit a broad endothermic consequence between about 160 and 280 °C, which is due to melting. The melting temperature of a polymer is usually associated with the peak temperature, nearly 258 °C in this case. In the temperature range between 350 and 500 °C, several overlapping endothermic and exothermic effects are observed in the DSC signal, which are due to the pyrolytic decomposition of the polymer content of the sample. The latter tin can be seen from the TG curve, which shows a mass loss pace of 66.3% in the same temperature range. A mass loss of one.3% was detected during the first heating, and another mass loss of two.3% occurred after switching to oxidative temper at college temperatures. The calculated residual mass of 30.1% matches with the nominal glass cobweb content of the sample. As decomposition temperature, the peak temperature of the calculated derivative of the TG curve, called DTG, is usually designated; it is 456 °C in the case of the sample "PA66-GF30_STA". In addition to peak temperatures, also extrapolated onset- and endset temperatures tin be evaluated according to known standards [24,25,26]; the size of a drinking glass transition is furthermore characterized by the step height ∆c p, and a melting effect by its area which is the enthalpy of melting (see Fig. 2).
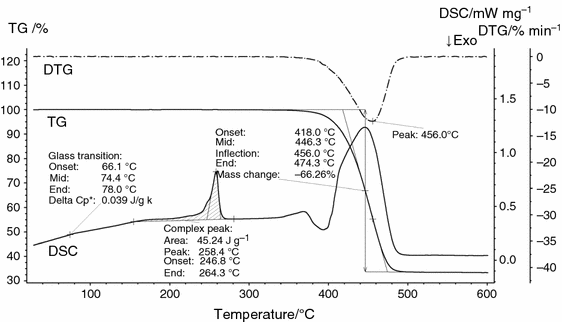
Temperature-dependent mass change (TG), the corresponding charge per unit of mass change (DTG) and the oestrus menstruation rate (DSC) of the polymer sample "PA66-GF30_STA". Not shown are the first heating to 300 °C and the higher temperature range (encounter text)
In order to evaluate the possibilities for material identification, the STA measurement displayed in Fig. two was analyzed by ways of Identify. The assay was restricted in the start instance to just the DSC bespeak in the temperature range of involvement below 300 °C where glass transition and melting occur in nigh polymers. The virtually similar database entries are summarized in a hitting list shown in Fig. 3a. It can exist seen that not but were polymers of type PA66 (polyamide 66) institute with high similarity values but besides ETFE (ethylene-tetrafluoroethylene), FEP (tetrafluoroethylene/hexafluoropropylene copolymer), PET (polyethylene terephthalate) and several other polymer types. This situation of multiple interpretations possible—and thus no definite material identification—is exactly what was expected from the literature data depicted in Fig. 1a. The DSC curves of the two nearly similar database entries are overlaid with the DSC curve of the polymer sample "PA66-GF30_STA" (see Fig. 3b). Visible are differences in the drinking glass transition and melting temperatures too as in the shape of the endothermic melting effects; there is evidently no perfect match betwixt the DSC curve of the sample "PA66-GF30_STA" and the database curves.
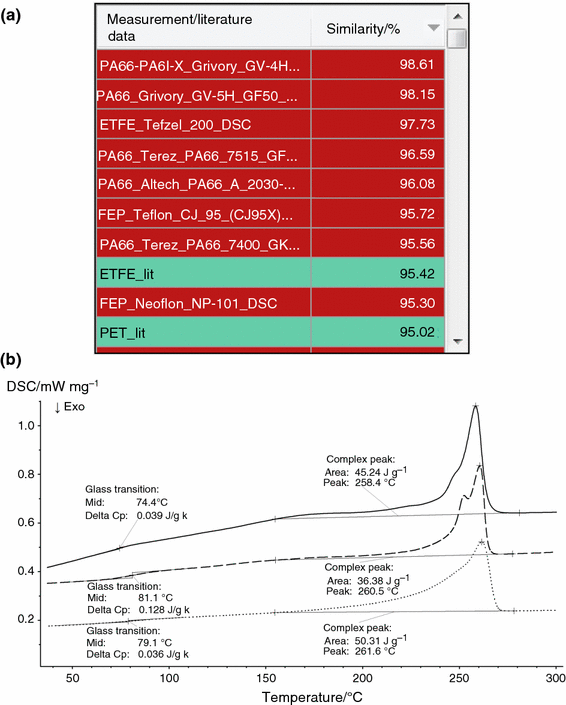
a Results from Place (striking list) for the STA measurement shown in Fig. 2 (sample "PA66-GF30_STA"). Just the DSC signal was considered, and the search temperature range was restricted to the range of 30–300 °C. The light-green colour refers to the library with NETZSCH literature information of almost 70 polymer types [21]; the cerise colour to entries of the KIMW [15] database containing DSC measurements on 600 different commercially available polymers [16]. b Temperature-dependent heat menstruation rate (DSC) of the polymer sample "PA66-GF30_STA" (measurement shown in Fig. two, solid line) in comparison with the DSC curves of the database entries "PA66-PA6I-X Grivory GV 4H GF40_DSC" (dashed line) and "ETFE Tefzel 200_DSC" (dotted line). The latter curves, which are shifted in the y direction for clarity, are selected Identify search results (a). (Color figure online)
Of course, comparisons between DSC measurements originating from different instruments, particularly from STA and stand-alone DSC devices, have to be considered advisedly. The glass transition may be less pronounced in an STA measurement due to lower DSC sensitivity and a greater impact of the baseline. Furthermore, temperature and sensitivity calibrations of different instruments and likewise the fourth dimension constants of the DSC sensors may differ. Such uncertainties are usual and have to be kept in mind for a database search. In the case of the example of Fig. iii, the algorithm of Identify was set to "qualitative", which disregards the size of the furnishings. This makes sense because a filled polymer was investigated. Still, it turns out that no other algorithm setting available improves the situation of multiple interpretations discussed in this case.
As a next stride, the TG information from the STA measurement of Fig. ii was besides analyzed using Identify, every bit is illustrated in Fig. 4a, b. The algorithm of Identify was over again set to "qualitative". The best hit, "PA66-GF30_TGA", is a TG database measurement on exactly the same material as that of the measurement of the sample "PA66-GF30_STA". Evidently, the database search revealed many farther TG curves of various polymer types which have also a high similarity to the TG curve of the polymer sample "PA66-GF30_STA" (see Fig. 4a, b)—which was again expected from literature data (see Fig. 1b). And this means that in the case of this example, the database search regarding the TG curve is again not definite—as was the instance for the corresponding DSC curve (see Fig. 3a, b).
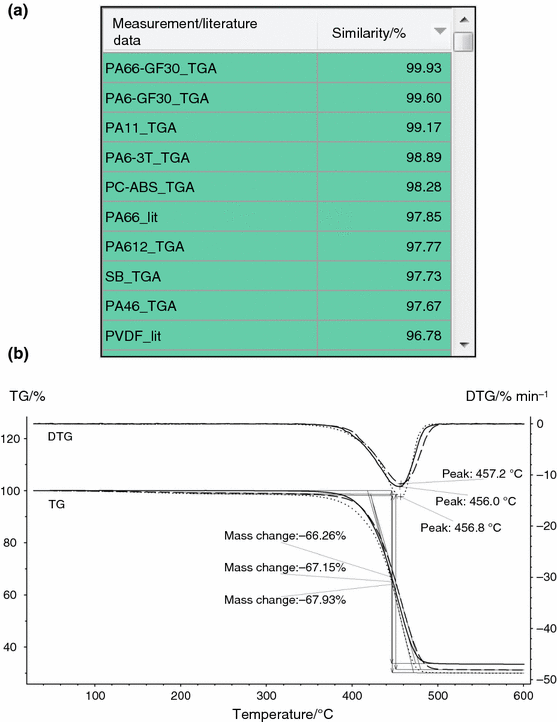
a Results from Place (striking listing) for the STA measurement shown in Fig. 2 (sample "PA66-GF30_STA"). Just the TG signal was considered, and the search temperature range was restricted to the range of 300–600 °C. The NETZSCH polymer libraries with measurements and literature data [21] of about lxx polymer types were used for the database search. b Temperature-dependent mass change (TG), the corresponding rate of mass modify (DTG) of the polymer sample "PA66-GF30_STA" (measurement shown in Fig. 2, solid line) in comparing with the TG curves of the database entries "PA66-GF30_TGA" (dashed line) and "PA6-GF30_TGA" (dotted line), which are selected Place search results (a)
The answer to this problem is the combination of the search results for the DSC and TG curves: The merely polymer type which revealed a high similarity to both the DSC and TG curves is PA66 (see Figs. 3a, 4a). In dissimilarity, polymer types like PA6 (polyamide half dozen) and PA11 (polyamide xi), which showed a high similarity to the TG curve (come across Fig. 4a), are unlikely because their similarity to the DSC bend is only below about fifty% and thus not visible from Fig. 3a. And from the other indicate of view, polymer types similar ETFE and PET, which are a reasonable possibility when regarding the DSC bend (see Fig. 3a), are likewise unlikely considering their similarity to the TG bend is relatively low (ETFE_TGA: 58.two%, PET_TGA: 91.3%). These findings can exist expected from the literature data shown in Fig. 1a, b.
In summary, the simultaneous measurement by TG and DSC in combination with the database search for both signal types by means of Identify leads to a clear identification and verification of the polymer type equally "PA66-GF30" with a relatively loftier certainty. In this example, the DSC and TG signals of an STA measurement were analyzed using Identify consecutively; this implies that exactly this kind of investigation is too possible based on ii independent measurements of the same type of sample performed on stand-alone DSC and TG instruments. In addition, Identify allows for simultaneous incorporation of DSC and TG signals into the database search which may originate from either two independent measurements or from one STA measurement. This should exist demonstrated using the library with literature data for about 70 polymer types [21]; a library containing STA measurements does not nonetheless exist, but the literature information practice contain information about glass transitions, melting and too decomposition temperatures in each individual database entry. As can be seen from Fig. 5, the assay of the unabridged STA measurement (DSC and TG at once) by means of Place consistently revealed PA66 as the all-time striking, while other polymer types were discriminated; the algorithm was again set to "qualitative", thus disregarding the size of all furnishings.

Results from Identify (hit listing) for the STA measurement shown in Fig. two (sample "PA66-GF30_STA"). Both the TG and DSC signals were considered simultaneously; the search temperature range was restricted to the range of xxx–600 °C. The library with NETZSCH literature data of about seventy polymer types [21] was used for the database search
For the sake of abyss, it should be noted once again that there are too polymers like PC (polycarbonate) that are purely amorphous and therefore only show a glass transition but no melting effect. And there are other polymers similar PTFE (polytetrafluoroethylene), where the glass transition exists just is typically not observable in the DSC signal considering the consequence is too weak. In both cases, which are not illustrated in Fig. 1, i of the effect types glass transition or melting is absent in the DSC indicate. Such polymers obviously distinguish themselves strongly from polymers that exhibit both effect types in the DSC signal. However, multiple interpretations are possible in comparison with other polymers that have in this case as well just a glass transition occurring in the aforementioned temperature range. An example would be the two polymer types PS (polystyrene) and PVC-U (polyvinylchloride without plasticizer), which both typically exhibit a glass transition in the range of 80–90 °C. In such a situation, the additional information from the TG betoken clearly helps to differentiate between the two polymers: In this example, PS shows only one decomposition footstep in the temperature range around 430 °C, whereas PVC-U exhibits two decomposition steps, the first around 300 °C and the second effectually 470 °C (DTG pinnacle temperatures) equally was depicted in Ref. [14].
Incorporation of c-DTA ® in add-on to TG
Unremarkably, the capabilities of TG instruments are restricted to the measurement of mass changes as a function of temperature or time. The TG musical instrument used for this work, yet, is able to also provide data regarding energetic furnishings in terms of the c-DTA ® signal. Compared to a true DSC point, c-DTA ® has a longer time abiding and is certainly less sensitive, which is a drawback especially for the detection of glass transitions. Secondly, c-DTA ® is only a semiquantitative bend without any enthalpy calibration. Nevertheless, the additional information from c-DTA ® can besides be incorporated for a definite identification of a sample material as was shown for TG-DSC higher up. Effigy 6a displays the results of a TG measurement of the sample "PB_TGA_new" including the c-DTA ® bend in the relevant temperature range. The latter reveals melting of the sample at a top temperature of about 122 °C; decomposition of the sample can exist seen from the mass loss in the temperature range between about 420 and 470 °C. In Fig. 6b, the results of Identify are depicted where TG and c-DTA ® were simultaneously incorporated into the database search; the algorithm was once again set to "qualitative", thus disregarding the size of all effects. The all-time hits, "PB_DSC" and "PB_TGA", are TG and DSC measurements on exactly the aforementioned polymer material as the sample "PB_TGA_new". There are polymer types which have a high similarity to the TG curve, but there is no other polymer type in the database where both the TG and DSC measurements have a high similarity to the measurement "PB_TGA_new" including its c-DTA ® curve. The definite verification of the polymer type Lead (polybutene) and the discrimination of other polymer types is once again demonstrated when the search library containing literature data including both, caloric effects and decomposition at the same time, are used (see Fig. 6c).
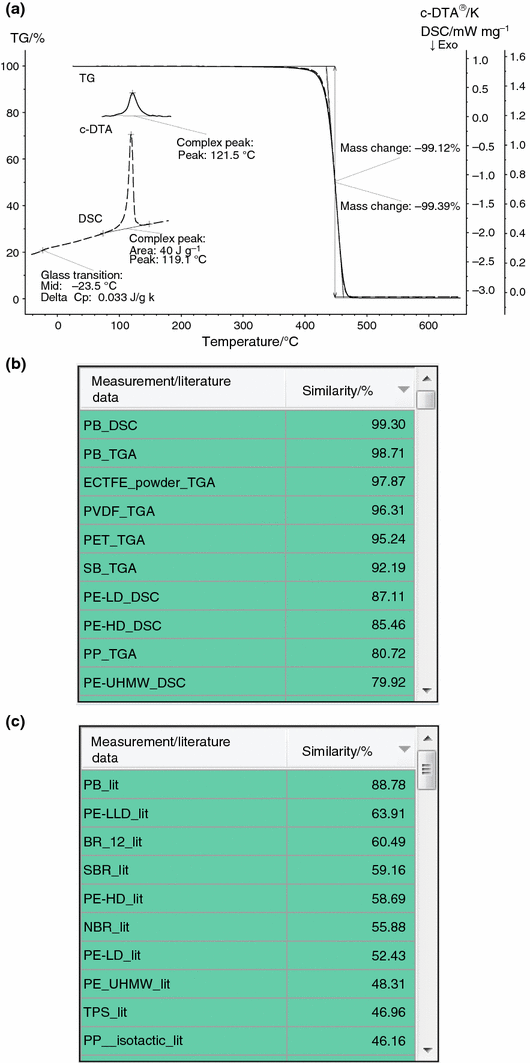
a Temperature-dependent mass change (TG) and c-DTA ® curve of the polymer sample "PB_TGA_new" (solid lines); not shown are the commencement heating to 160 °C and the college temperature range where no meaning mass changes occurred. For comparison, the TG and DSC curves of the database entries "PB_TGA" and "PB_DSC" (dashed lines) are shown, which are selected Identify search results (b). b Results from Identify (hit list) for the TG measurement shown in a (sample "PB_TGA_new"). Both, the TG and c-DTA ® curves were considered simultaneously; the search temperature range was restricted to the range of 30–600 °C. The NETZSCH library with measurements of about 70 polymer types [21] was used for the database search. c Results from Place (hit list) for the TG measurement shown in a (sample "PB_TGA_new"). Both, the TG and c-DTA ® curves were considered simultaneously; the search temperature range was restricted to the range of 30–600 °C. The NETZSCH library with literature data of about seventy polymer types [21] was used for the database search
Incorporation of curve specifics in improver to the main feature temperatures
Fortunately, the chief characteristic temperatures (glass transition, melting and decomposition temperatures) are not the just information that can exist extracted from DSC and TG curves. Drinking glass transitions and melting furnishings occurring in the DSC signal differ in size and broadness and in some cases also exhibit specific shapes for the private polymer types. In addition, fifty-fifty polymer types with i-stride decomposition in some cases exhibit preceding effects and afterwards-effects which betoken to mass loss before and afterward the main decomposition step. The broadness and the size of the main decomposition step can likewise vary among unlike polymer types. All those properties can be partially considered past Place depending on the algorithm settings; the drinking glass transition, melting and decomposition temperatures are nonetheless the well-nigh important values. The following example should demonstrate in particular the benefit of the additional information that is gained past just overlaying the bodily measurement with database curves and visually comparing the curves [16]. A DSC measurement of the commercially bachelor polymer "PA6 Durethan BKV30 H2.0" was analyzed past means of Place using its standard algorithm settings, which take into account all properties of the effects occurring in the DSC curve. Exactly the same measurement is also present in the KIMW database and thus appears as the best striking (encounter Fig. 7a). From the resulting striking lists, it can furthermore exist seen that many similar PA6 measurements were constitute, but also the two database measurements of PA610. The slightly unlike characteristic temperatures, especially the glass transition temperature (come across Fig. 7b), already lead to a significant lowering of the similarity between the PA610 measurements and many of the PA6 database entries, and thus also lead to a discrimination between PA610 and the PA6 measurement investigated. The PA610 curves furthermore exhibit a pronounced shoulder effectually 170 °C and an boosted small melting summit at about 210 °C which do not occur in the PA6 measurements, as can be seen from the overlay of the corresponding DSC curves shown in Fig. 7b. Another finding is that the entire PA6 class containing 39 measurements on different PA6 grades has a lower mean similarity than the PA610 form, which contains but two measurements (encounter Fig. 7a). This is due to the fact that for some PA6 grades, an boosted endothermic effect occurs at around 110 °C, which also leads to differentiation amidst the PA6 database entries (see Fig. 7b). Finally, it can be recognized from the Place results that the similarity of the classes of all other polymer types nowadays in the database is significantly lower than for PA6 and PA610, respectively, which likewise demonstrates the possibility of differentiation of many polymer types (meet Fig. 7a). A prerequisite for these detailed results is of course the being of the large KIMW database [15, 16].
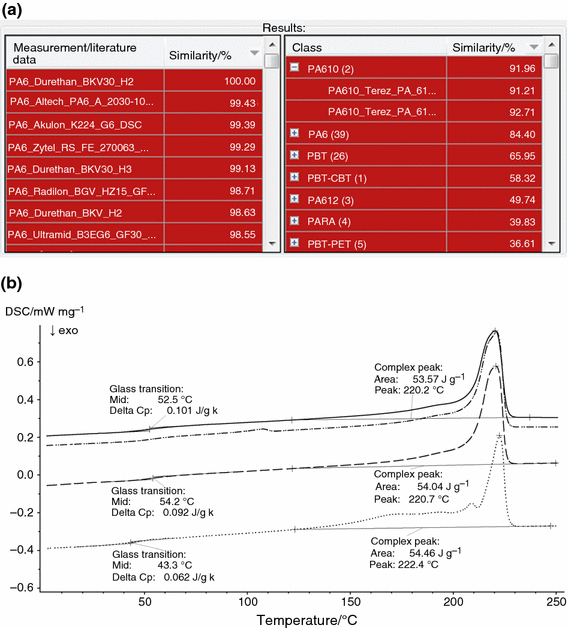
a Results from Identify (hit lists) regarding a DSC measurement of the polymer "PA6 Durethan BKV30 H2.0". The KIMW [15] database containing DSC measurements on 600 dissimilar commercially available polymers [sixteen] was used for the search. b Temperature-dependent rut menstruation charge per unit (DSC) of the polymer sample "PA6 Durethan BKV30 H2.0" (solid line) in comparison with the DSC curves of the database entries "PA6 Altech PA6 A 2030-109 GF30" (dashed line) and "PA610 Terez PA6.ten GF30 H ECO" (dotted line). The latter curves are selected Identify search results (a). The exemplary database curve of the Polymer "PA6 Schulamid 6 GF30 H" is furthermore shown (dashed-dotted line, see text). All curves are shifted in the y direction for clarity
Conclusions
The main topic of this work is the identification of polymers by ways of differential scanning calorimetry (DSC), thermogravimetry (TG) and simultaneous thermal analysis (STA) in combination with a computer-assisted database search using the Identify software. The identification can more often than not be carried out mainly on the ground of characteristic caloric effects of the type glass transition, crystallization or melting and on the footing of mass changes, which reflect the temperature-dependent decomposition of a sample. In general, a single measurement can unfortunately be interpreted in multiple ways limiting this method of fabric identification [14]. This difficulty is considerably minimized past the consecutive or simultaneous incorporation of ii types of measurement signals into the database search: TG and DSC or TG and c-DTA ® . As application examples, a definite identification of polymers of type PA66 (polyamide 66) and PB (polybutene) was demonstrated.
The KIMW [fifteen] library available for Place was furthermore used, which contains DSC curves for 600 unlike commercially available polymers (nigh 130 polymer types)—besides including data about trade names, colors and filler contents [16]. This large database allows for more distinct polymer identification; this is due to the multitude of polymer types present in the database just likewise due to the availability of several different polymers of the same blazon which may showroom pregnant differences in their DSC curves. The differentiation betwixt PA6 (polyamide 6) and PA610 (polyamide 610) was depicted as an example.
References
-
Ehrenstein GW, Riedel 1000, Trawiel P. Thermal analysis of plastics: theory and practice. Cincinnati: Hanser Gardner Publications; 2004.
-
Frick A, Stern C. DSC-Prüfung in der Anwendung. München: Carl Hanser Verlag; 2013.
-
Mitchell Yard, Fenalla F, Nordon A, Leung Tang P, Gibson LT. Assessment of historical polymers using adulterate total reflectance-Fourier transform infra-cerise spectroscopy with principal component assay. Herit Sci. 2013;. doi:x.1186/2050-7445-1-28.
-
Hemminger WF, Cammenga HK. Methoden der thermischen analyse. Heidelberg: Springer; 1989.
-
Schmölzer Southward. Temperature Taken. Kunstst Int. 2009;10:55–7.
-
ASTM Eastward 473-16, standard terminology related to thermal assay and rheology.
-
Opfermann J, Schmidt M. Verfahren zur Durchführung der Differential-Thermoanalyse. Deutsches Patent- und Markenamt. 2004. DE 199 34 448 B4 2004.09.30.
-
Denner T, et al. Method for performing a differential thermal analysis. United states Patent. 2014. US 2014/0204971.
-
Schindler A, Neumann G, Rager A, Füglein E, Denner T, Blumm J. A novel direct coupling of simultaneous thermal analysis (STA) and Fourier transform-infrared (FT-IR) spectroscopy. J Therm Anal Calorim. 2013;113:1091–102.
-
Schindler A. Materialerkennung und Qualitätskontrolle: Auswerten, identifizieren und interpretieren. Plastverarbeiter. 2014;01:xxx–2.
-
Schindler A. AutoEvaluation – Automatische Auswertung von DSC-Kurven. Identify – Das neue DSC-Kurvenerkennungs- und Datenbanksystem. Presentation during annual symposium of the AK Thermophysik (part of the GEFTA). 2014; March 17–18.
-
Schindler A. Automatic evaluation and identification of DSC curves. Plast Eng. 2014. http://www.plasticsengineering.org/ProductFocus/productfocus.aspx?ItemNumber=20498.
-
Moukhina E, Schindler A. Automatic evaluation and identification of DSC curves. Presentation during international GEFTA symposium "thermal analysis and calorimetry in industry and inquiry". 2014; September 16–19.
-
Schindler A, Strasser C, Schmölzer S, Bodek M, Seniuta R, Wang Ten. Database-supported thermal analysis involving automatic evaluation, identification and classification of measurement curves. J Therm Anal Calorim. 2016. doi:10.1007/s10973-015-5026-x.
-
KIMW Prüf- und Analyse GmbH, Karolinenstraße viii, D-58507 Lüdenscheid, Germany.
-
Doedt M, Schindler A, Pflock T. DSC-Auswertung mit einem Klick - Datenbank-Integration und Evaluationssoftware vereinfachen Polymeridentifizierung. Kunststoffe. 2016;10:189–91.
-
Fueglein E, Kaisersberger E. About the development of databases in thermal analysis. J Therm Anal Calorim. 2015;. doi:ten.1007/s10973-014-4381-3.
-
Fueglein E. Most the use of identify—a thermoanalytical database—for characterization and nomenclature of recycled polyamides. J Therm Anal Calorim. 2015;. doi:10.1007/s10973-015-4583-three.
-
http://www.campusplastics.com/.
-
http://www.yard-base.de/de/produkte/materialdatacenter.html.
-
Thermal properties of elements, polymers, alloys, ceramics. www.netzsch.com/TPoE, www.netzsch.com/TPoP, www.netzsch.com/TPoA, world wide web.netzsch.com/TPoC.
-
Lee WA, Knight GJ. Ratio of the glass transition temperature to the melting point in polymers. Br Polym J. 1970;2:73–80.
-
Schindler A, Moukhina E, Pflock T. Automated identification and classification of thermoplastic elastomers by means of DSC and TGA. TPE Mag. 2016;8:188–91.
-
ASTM D3418-15. Standard exam method for transition temperatures and enthalpies of fusion and crystallization of polymers by differential scanning calorimetry.
-
ISO 11357-1:2009. Plastics—differential scanning calorimetry (DSC)—office ane: general principles.
-
ISO 11358-i:2014. Thermogravimetry (TG) of polymers—role one: general principles.
Author data
Affiliations
Corresponding author
Rights and permissions
Open up Access This article is distributed nether the terms of the Creative Eatables Attribution 4.0 International License (http://creativecommons.org/licenses/by/iv.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided y'all give appropriate credit to the original writer(s) and the source, provide a link to the Creative Eatables license, and indicate if changes were made.
Reprints and Permissions
About this commodity
Cite this article
Schindler, A., Doedt, M., Gezgin, Ş. et al. Identification of polymers by ways of DSC, TG, STA and calculator-assisted database search. J Therm Anal Calorim 129, 833–842 (2017). https://doi.org/10.1007/s10973-017-6208-5
-
Received:
-
Accepted:
-
Published:
-
Issue Appointment:
-
DOI : https://doi.org/10.1007/s10973-017-6208-5
Keywords
- Identification
- Polymers
- Database
- DSC
- TG
- STA
- c-DTA ®
Source: https://link.springer.com/article/10.1007/s10973-017-6208-5
0 Response to "How to Read a Dsc Curve Plastic"
Post a Comment